-
CETANIL CT TAB 10`S ₦3,074.50 QTY: 1
Customer matched zone "Lagos Delivery Options"
“BIOVAS 40MG TAB 10`S” has been added to your basket. Continue shopping
Sort by:
194481–194496 of 371823 Results
-
-
-
SaleLiglimet 500 2.5 mg+500 mgThis is indicated as an adjunct to diet and exercise to improve glycemic control in adults with type 2 diabetes mellitus when treatment with both Linagliptin and Metformin Hydrochloride is appropriateTheropeutic ClassCombination Oral hypoglycemic preparationsPharmacologyLinagliptin is indicated to improve glycemic control in patients with type 2 diabetes mellitus. Linagliptin is an inhibitor of DPP-4 (dipeptidyl peptidase-4), an enzyme that degrades the incretin hormones GLP-1 (glucagon like peptide-1) and GIP (glucose dependent insulinotropic polypeptide). Thus, Linagliptin increases the concentrations of active incretin hormones, stimulating the release of insulin from pancreatic beta (?) cells in a glucose-dependent manner and decreasing the secretion of glucagon from pancreatic alpha (?) cells in the circulation.Metformin Hydrochloride is a biguanide type oral antihyperglycemic drug used in the management of type 2 diabetes. It lowers both basal and postprandial plasma glucose. Its mechanism of action is different from those of sulfonylureas and it does not produce hypoglycemia. Metformin Hydrochloride decreases hepatic glucose production, decreases intestinal absorption of glucose and improves insulin sensitivity by an increase in peripheral glucose uptake and utilization.Dosage & Administration of Liglimet 500 2.5 mg+500 mgLinagliptin & Metformin immediate release tablet: The dosage of Linagliptin & Metformin should be individualized on the basis of both effectiveness and tolerability. Maximum recommended dose of 2.5 mg Linagliptin and 1000 mg Metformin Hydrochloride twice daily with meals. Dose escalation should be gradual to reduce the gastrointestinal (GI) side effects associated with Metformin Hydrochloride use.Recommended starting dose: In patients currently not treated with Metformin Hydrochloride, initiate treatment with 2.5 mg Linagliptin and 500 mg Metformin Hydrochloride twice daily. In patients already treated with Metformin Hydrochloride, start with 2.5 mg Linagliptin and the current dose of Metformin Hydrochloride twice daily. Patients already treated with Linagliptin and Metformin Hydrochloride, individual components may be switched to this combination containing the same doses of each component.Linagliptin & Metformin extend release tablet: The dosage of this combination should be individualized on the basis of both effectiveness and tolerability, while not exceeding the maximum recommended total daily dose of Linagliptin 5 mg and Metformin Hydrochloride 2000 mg. this combination should be given once daily with a meal.Recommended starting dose: In patients currently not treated with metformin, initiate this combination treatment with 5 mg Linagliptin/1000 mg Metformin Hydrochloride extended-release once daily with a meal.In patients already treated with Metformin, start this combination with 5 mg of Linagliptin total daily dose and a similar total daily dose of Metformin once daily with a meal.In patients already treated with Linagliptin & Metformin immediate release tablet, switch to extend release tablet containing 5 mg of Linagliptin total daily dose and a similar total daily dose of Metformin once daily with a meal.5 mg Linagliptin & 1000 mg Metformin Hydrochloride extended-release tablet should be taken as a single tablet once daily. Patients using 2.5 mg Linagliptin & 1000 mg Metformin extended release tablets should take two tablets together once daily.Dosage of Liglimet 500 2.5 mg+500 mgLinagliptin & Metformin immediate release tablet: The dosage of Linagliptin & Metformin should be individualized on the basis of both effectiveness and tolerability. Maximum recommended dose of 2.5 mg Linagliptin and 1000 mg Metformin Hydrochloride twice daily with meals. Dose escalation should be gradual to reduce the gastrointestinal (GI) side effects associated with Metformin Hydrochloride use.Recommended starting dose: In patients currently not treated with Metformin Hydrochloride, initiate treatment with 2.5 mg Linagliptin and 500 mg Metformin Hydrochloride twice daily. In patients already treated with Metformin Hydrochloride, start with 2.5 mg Linagliptin and the current dose of Metformin Hydrochloride twice daily. Patients already treated with Linagliptin and Metformin Hydrochloride, individual components may be switched to this combination containing the same doses of each component.Linagliptin & Metformin extend release tablet: The dosage of this combination should be individualized on the basis of both effectiveness and tolerability, while not exceeding the maximum recommended total daily dose of Linagliptin 5 mg and Metformin Hydrochloride 2000 mg. this combination should be given once daily with a meal.Recommended starting dose: In patients currently not treated with metformin, initiate this combination treatment with 5 mg Linagliptin/1000 mg Metformin Hydrochloride extended-release once daily with a meal.In patients already treated with Metformin, start this combination with 5 mg of Linagliptin total daily dose and a similar total daily dose of Metformin once daily with a meal.In patients already treated with Linagliptin & Metformin immediate release tablet, switch to extend release tablet containing 5 mg of Linagliptin total daily dose and a similar total daily dose of Metformin once daily with a meal.5 mg Linagliptin & 1000 mg Metformin Hydrochloride extended-release tablet should be taken as a single tablet once daily. Patients using 2.5 mg Linagliptin & 1000 mg Metformin extended release tablets should take two tablets together once daily.Interaction of Liglimet 500 2.5 mg+500 mgCationic drugs (amiloride, digoxin, morphine, ranitidine, trimethoprim etc.): May reduce metformin elimination. P-glycoprotien/CYP3A4 inducer (i.e. rifampin): The efficacy of this medicine may be reduced when administered in combination.ContraindicationsAlthough Linagliptin undergoes minimal renal excretion, Metformin Hydrochloride is known to be substantially excreted by the kidney. The risk of Metformin Hydrochloride accumulation and lactic acidosis increases with the degree of renal impairment. Therefore, this combination is contraindicated in patients with renal impairment. It is also contraindicated in acute or chronic metabolic acidosis (diabetic ketoacidosis) and in hypersensitivity to Linagliptin or Metformin Hydrochloride.Side Effects of Liglimet 500 2.5 mg+500 mgMost common side effects are nasopharyngitis and diarrhea. Hypoglycemia is more common in patients treated with this combination and sulfonylureas.Pregnancy & LactationThere are no adequate and well-controlled studies in pregnant women with this combination or its individual component; so it should be used during pregnancy only if clearly needed. Caution should also be excercised when it is administered to a lactating mother.Precautions & WarningsIn a patient with lactic acidosis who is taking Metformin, the drug should be discontinued immediately and supportive therapy promptly instituted. There have been postmarketing reports of acute pancreatitis. If pancreatitis is suspected, promptly discontinue Linagliptin & Metformin. Temporarily discontinue Linagliptin & Metformin?in patients undergoing radiologic studies with intravascular administration of iodinated contrast materials or any surgical procedures necessitating restricted intake of food and fluids. Metformin may lower Vitamin B12 levels; so hematologic parameters shoud be monitored annually.Overdose Effects of Liglimet 500 2.5 mg+500 mgIn the event of an overdose with this combination the usual supportive measures (i.e. remove unabsorbed material from the gastrointestinal tract, perform clinical monitoring, and institute supportive treatment) should be employed. Removal of Linagliptin by hemodialysis or peritoneal dialysis is unlikely but Metformin Hydrochloride is dialyzable. During controlled clinical trials in healthy subjects, with single doses of up to 600 mg of Linagliptin (equivalent to 120 times the recommended daily dose), there were no dose-related clinical adverse drug reactions. Overdose of Metformin Hydrochloride has occurred in case of ingestion of amounts greater than 50 grams. Hypoglycemia was reported in approximately 10% of cases, but no causal association with Metformin Hydrochloride has been established. Lactic acidosis has been reported in approximately 32% of Metformin Hydrochloride overdose cases.Storage ConditionsKeep in a cool & dry place (below 30?C), protected from light & moisture. Keep out of the reach of children.Drug ClassesCombination Oral hypoglycemic preparationsMode Of ActionLinagliptin is indicated to improve glycemic control in patients with type 2 diabetes mellitus. Linagliptin is an inhibitor of DPP-4 (dipeptidyl peptidase-4), an enzyme that degrades the incretin hormones GLP-1 (glucagon like peptide-1) and GIP (glucose dependent insulinotropic polypeptide). Thus, Linagliptin increases the concentrations of active incretin hormones, stimulating the release of insulin from pancreatic beta (?) cells in a glucose-dependent manner and decreasing the secretion of glucagon from pancreatic alpha (?) cells in the circulation.Metformin Hydrochloride is a biguanide type oral antihyperglycemic drug used in the management of type 2 diabetes. It lowers both basal and postprandial plasma glucose. Its mechanism of action is different from those of sulfonylureas and it does not produce hypoglycemia. Metformin Hydrochloride decreases hepatic glucose production, decreases intestinal absorption of glucose and improves insulin sensitivity by an increase in peripheral glucose uptake and utilization.PregnancyThere are no adequate and well-controlled studies in pregnant women with this combination or its individual component; so it should be used during pregnancy only if clearly needed. Caution should also be excercised when it is administered to a lactating mother.Sku: 1736105253-3798
Liglimet 5002.5 mg+500 mg
₦4,290.00Original price was: ₦4,290.00.₦3,861.00Current price is: ₦3,861.00.₦4,290.00Original price was: ₦4,290.00.₦3,861.00Current price is: ₦3,861.00. Add to basket Quick View -
SaleLiglimet 850 2.5 mg+850 mgThis is indicated as an adjunct to diet and exercise to improve glycemic control in adults with type 2 diabetes mellitus when treatment with both Linagliptin and Metformin Hydrochloride is appropriateTheropeutic ClassCombination Oral hypoglycemic preparationsPharmacologyLinagliptin is indicated to improve glycemic control in patients with type 2 diabetes mellitus. Linagliptin is an inhibitor of DPP-4 (dipeptidyl peptidase-4), an enzyme that degrades the incretin hormones GLP-1 (glucagon like peptide-1) and GIP (glucose dependent insulinotropic polypeptide). Thus, Linagliptin increases the concentrations of active incretin hormones, stimulating the release of insulin from pancreatic beta (?) cells in a glucose-dependent manner and decreasing the secretion of glucagon from pancreatic alpha (?) cells in the circulation.Metformin Hydrochloride is a biguanide type oral antihyperglycemic drug used in the management of type 2 diabetes. It lowers both basal and postprandial plasma glucose. Its mechanism of action is different from those of sulfonylureas and it does not produce hypoglycemia. Metformin Hydrochloride decreases hepatic glucose production, decreases intestinal absorption of glucose and improves insulin sensitivity by an increase in peripheral glucose uptake and utilization.Dosage & Administration of Liglimet 850 2.5 mg+850 mgLinagliptin & Metformin immediate release tablet: The dosage of Linagliptin & Metformin should be individualized on the basis of both effectiveness and tolerability. Maximum recommended dose of 2.5 mg Linagliptin and 1000 mg Metformin Hydrochloride twice daily with meals. Dose escalation should be gradual to reduce the gastrointestinal (GI) side effects associated with Metformin Hydrochloride use.Recommended starting dose: In patients currently not treated with Metformin Hydrochloride, initiate treatment with 2.5 mg Linagliptin and 500 mg Metformin Hydrochloride twice daily. In patients already treated with Metformin Hydrochloride, start with 2.5 mg Linagliptin and the current dose of Metformin Hydrochloride twice daily. Patients already treated with Linagliptin and Metformin Hydrochloride, individual components may be switched to this combination containing the same doses of each component.Linagliptin & Metformin extend release tablet: The dosage of this combination should be individualized on the basis of both effectiveness and tolerability, while not exceeding the maximum recommended total daily dose of Linagliptin 5 mg and Metformin Hydrochloride 2000 mg. this combination should be given once daily with a meal.Recommended starting dose: In patients currently not treated with metformin, initiate this combination treatment with 5 mg Linagliptin/1000 mg Metformin Hydrochloride extended-release once daily with a meal.In patients already treated with Metformin, start this combination with 5 mg of Linagliptin total daily dose and a similar total daily dose of Metformin once daily with a meal.In patients already treated with Linagliptin & Metformin immediate release tablet, switch to extend release tablet containing 5 mg of Linagliptin total daily dose and a similar total daily dose of Metformin once daily with a meal.5 mg Linagliptin & 1000 mg Metformin Hydrochloride extended-release tablet should be taken as a single tablet once daily. Patients using 2.5 mg Linagliptin & 1000 mg Metformin extended release tablets should take two tablets together once daily.Dosage of Liglimet 850 2.5 mg+850 mgLinagliptin & Metformin immediate release tablet: The dosage of Linagliptin & Metformin should be individualized on the basis of both effectiveness and tolerability. Maximum recommended dose of 2.5 mg Linagliptin and 1000 mg Metformin Hydrochloride twice daily with meals. Dose escalation should be gradual to reduce the gastrointestinal (GI) side effects associated with Metformin Hydrochloride use.Recommended starting dose: In patients currently not treated with Metformin Hydrochloride, initiate treatment with 2.5 mg Linagliptin and 500 mg Metformin Hydrochloride twice daily. In patients already treated with Metformin Hydrochloride, start with 2.5 mg Linagliptin and the current dose of Metformin Hydrochloride twice daily. Patients already treated with Linagliptin and Metformin Hydrochloride, individual components may be switched to this combination containing the same doses of each component.Linagliptin & Metformin extend release tablet: The dosage of this combination should be individualized on the basis of both effectiveness and tolerability, while not exceeding the maximum recommended total daily dose of Linagliptin 5 mg and Metformin Hydrochloride 2000 mg. this combination should be given once daily with a meal.Recommended starting dose: In patients currently not treated with metformin, initiate this combination treatment with 5 mg Linagliptin/1000 mg Metformin Hydrochloride extended-release once daily with a meal.In patients already treated with Metformin, start this combination with 5 mg of Linagliptin total daily dose and a similar total daily dose of Metformin once daily with a meal.In patients already treated with Linagliptin & Metformin immediate release tablet, switch to extend release tablet containing 5 mg of Linagliptin total daily dose and a similar total daily dose of Metformin once daily with a meal.5 mg Linagliptin & 1000 mg Metformin Hydrochloride extended-release tablet should be taken as a single tablet once daily. Patients using 2.5 mg Linagliptin & 1000 mg Metformin extended release tablets should take two tablets together once daily.Interaction of Liglimet 850 2.5 mg+850 mgCationic drugs (amiloride, digoxin, morphine, ranitidine, trimethoprim etc.): May reduce metformin elimination. P-glycoprotien/CYP3A4 inducer (i.e. rifampin): The efficacy of this medicine may be reduced when administered in combination.ContraindicationsAlthough Linagliptin undergoes minimal renal excretion, Metformin Hydrochloride is known to be substantially excreted by the kidney. The risk of Metformin Hydrochloride accumulation and lactic acidosis increases with the degree of renal impairment. Therefore, this combination is contraindicated in patients with renal impairment. It is also contraindicated in acute or chronic metabolic acidosis (diabetic ketoacidosis) and in hypersensitivity to Linagliptin or Metformin Hydrochloride.Side Effects of Liglimet 850 2.5 mg+850 mgMost common side effects are nasopharyngitis and diarrhea. Hypoglycemia is more common in patients treated with this combination and sulfonylureas.Pregnancy & LactationThere are no adequate and well-controlled studies in pregnant women with this combination or its individual component; so it should be used during pregnancy only if clearly needed. Caution should also be excercised when it is administered to a lactating mother.Precautions & WarningsIn a patient with lactic acidosis who is taking Metformin, the drug should be discontinued immediately and supportive therapy promptly instituted. There have been postmarketing reports of acute pancreatitis. If pancreatitis is suspected, promptly discontinue Linagliptin & Metformin. Temporarily discontinue Linagliptin & Metformin?in patients undergoing radiologic studies with intravascular administration of iodinated contrast materials or any surgical procedures necessitating restricted intake of food and fluids. Metformin may lower Vitamin B12 levels; so hematologic parameters shoud be monitored annually.Overdose Effects of Liglimet 850 2.5 mg+850 mgIn the event of an overdose with this combination the usual supportive measures (i.e. remove unabsorbed material from the gastrointestinal tract, perform clinical monitoring, and institute supportive treatment) should be employed. Removal of Linagliptin by hemodialysis or peritoneal dialysis is unlikely but Metformin Hydrochloride is dialyzable. During controlled clinical trials in healthy subjects, with single doses of up to 600 mg of Linagliptin (equivalent to 120 times the recommended daily dose), there were no dose-related clinical adverse drug reactions. Overdose of Metformin Hydrochloride has occurred in case of ingestion of amounts greater than 50 grams. Hypoglycemia was reported in approximately 10% of cases, but no causal association with Metformin Hydrochloride has been established. Lactic acidosis has been reported in approximately 32% of Metformin Hydrochloride overdose cases.Storage ConditionsKeep in a cool & dry place (below 30?C), protected from light & moisture. Keep out of the reach of children.Drug ClassesCombination Oral hypoglycemic preparationsMode Of ActionLinagliptin is indicated to improve glycemic control in patients with type 2 diabetes mellitus. Linagliptin is an inhibitor of DPP-4 (dipeptidyl peptidase-4), an enzyme that degrades the incretin hormones GLP-1 (glucagon like peptide-1) and GIP (glucose dependent insulinotropic polypeptide). Thus, Linagliptin increases the concentrations of active incretin hormones, stimulating the release of insulin from pancreatic beta (?) cells in a glucose-dependent manner and decreasing the secretion of glucagon from pancreatic alpha (?) cells in the circulation.Metformin Hydrochloride is a biguanide type oral antihyperglycemic drug used in the management of type 2 diabetes. It lowers both basal and postprandial plasma glucose. Its mechanism of action is different from those of sulfonylureas and it does not produce hypoglycemia. Metformin Hydrochloride decreases hepatic glucose production, decreases intestinal absorption of glucose and improves insulin sensitivity by an increase in peripheral glucose uptake and utilization.PregnancyThere are no adequate and well-controlled studies in pregnant women with this combination or its individual component; so it should be used during pregnancy only if clearly needed. Caution should also be excercised when it is administered to a lactating mother.Sku: 1736104572-3595
Liglimet 8502.5 mg+850 mg
₦4,950.00Original price was: ₦4,950.00.₦4,455.00Current price is: ₦4,455.00.₦4,950.00Original price was: ₦4,950.00.₦4,455.00Current price is: ₦4,455.00. Add to basket Quick View -
-
LIGNEL HEALTHCARE VITAMIN D3 25MCG (1000IU)Sku: 1712561056-8031
LIGNEL HEALTHCARE VITAMIN D3 25MCG (1000IU)
₦8,300.00 -
LIGNEL HEALTHCARE VITAMIN D3 25MCG (5000IU)Sku: 1712561059-8032
LIGNEL HEALTHCARE VITAMIN D3 25MCG (5000IU)
₦10,500.00 -
Lignel Vitamin D3 25Mcg(1000Iu) Softgels X60Sku: 1746436362-730
Lignel Vitamin D3 25Mcg(1000Iu) Softgels X60
₦18,590.00 -
Lignel Vitamin D3 5000Iu Softgels x60Sku: 1746524533-46
Lignel Vitamin D3 5000Iu Softgels x60
₦18,850.00 -
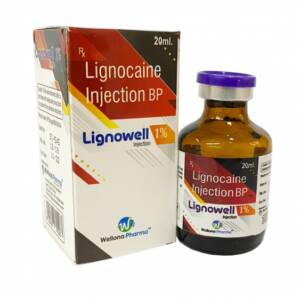
Lignocaine 1% Injection Trade Name: Lidowell, Lignowell Available Strength : 1%, 2% Available Combination : Lidocaine + Adrenaline, Lidocaine + Epinephrine Packing : 2 ml, 20 ml, 30 ml Pack Insert/Leaflet : Yes Therapeutic use : Anaesthetic Production Capacity : 1 million injection/monthSku: 1714922812-210
Lignocaine 1% Injection
₦100.00 -
-
-
-
-
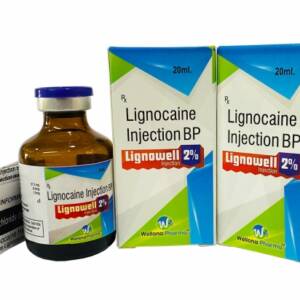
Lignocaine 2% Injection Trade Name: Lidowell, Lignowell Available Strength : 1%, 2% Packing : 2 ml, 20 ml, 30 ml Pack Insert/Leaflet : Yes Therapeutic use : Anaesthetic Production Capacity : 1 million injection/monthSku: 1714922809-209
Lignocaine 2% Injection
₦100.00 -
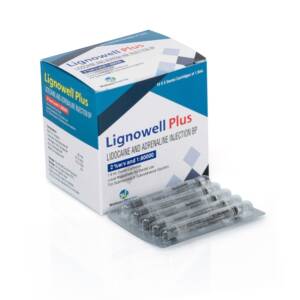
Lignocaine Adrenaline Dental Cartridges Trade Name: Lignowell Plus Available Strength : 1:80000 Packing : Cartridge Pack Insert/Leaflet : Yes Therapeutic use : Generic Drugs,Anaesthetic Production Capacity : 1 million injection/monthSku: 1714922803-208
Lignocaine Adrenaline Dental Cartridges
₦100.00